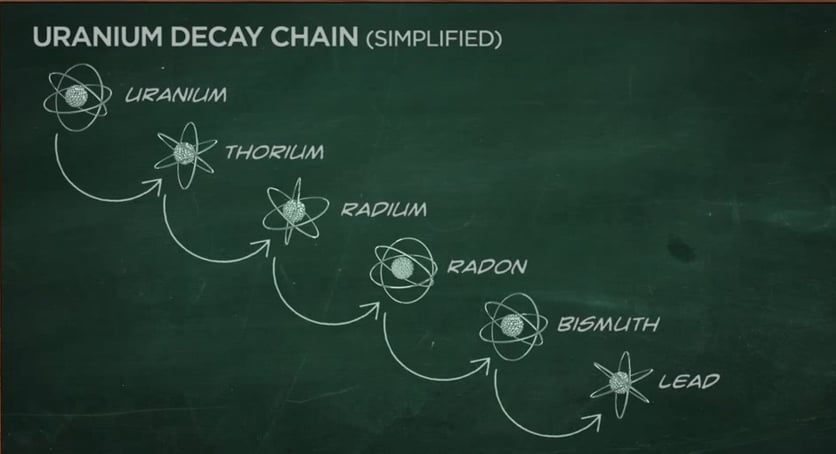
Energy is released as a result. Red spheres are neutrons, and blue spheres are protons. This produces U-236, which is unstable and splits into fission fragments and additional neutrons. Thus, the nuclear fission observed by Nishina and Kimura is highly symmetric. This could be taken as a suggestion that geologic storage of radioactive waste is feasible.\): Fission of uranium-235 ( 235U) can be induced by bombarding it with a neutron. An uranium isotope having a half-life of 6.5 d was unknown at that time. One of the interesting observations was that the bulk of the fission products seemed to be still in place in their geologic depository after nearly 2 billion years. 
It is presumed that ground water seeping through the ore served as a natural moderator to slow down the fission neutrons. Currently, uranium-235 (235 U an isotope of uranium with an atomic mass of 235) is currently used as fuel for nuclear fission reactions. One of the interesting observations was that the bulk of. radioactive decay series for thorium-232, uranium-235 and uranium-238. For U-235 (halflife 700 million years) and U-238 (halflife 4.5 billion years), this would give a concentration of about 3 for the U-235 at the time of the reaction. The heat energy released when nuclear fission can be used to generate electricity. These neutrons may induce a nuclear fission chain reaction if there is enough. Most radioactive forms of iodine are produced as a byproduct of a fission fission The splitting of an atomic nucleus into at least two other nuclei with the release of a relatively large. of iodine, and only one is not radioactive (Iodine-127). nucleus such as the isotope uranium 233, uranium 235, or plutonium 239. For example, uranium has thirty-seven different isotopes, including uranium-235 and uranium-238. Its specific activity is very low 2.2×10-6 Ci/g. There is no specific relationship between half-life and nuclear fission except. Uranium 235 occasionally decays by spontaneous fission with very low probability of 0.0000000072. The half-life of uranium 235 is 7 x 108 years. Because the half-life is different with U238 and U235, the higher the percentage of U235 so retroactively. Current ratio of U235 in natural uranium is the same anywhere in the solar system. For U-235 (halflife 700 million years) and U-238 (halflife 4.5 billion years), this would give a concentration of about 3% for the U-235 at the time of the reaction. As with natural radioactive decay, induced nuclear fission reactions release energy. Uranium 235 decays via alpha decay (by way of thorium-231) into 231 Pa. Calculates the dating in the past from the ratio of Uranium 235 in natural uranium. The age estimate from cores in the reactor zones suggest a time frame between 1.7 and 1.9 billion years ago. 
Other isotopes that can be induced to fission like this are plutonium-239, uranium-233, and thorium-232. For all practical purposes, plutonium-239 does not. The most important fissile materials for nuclear energy and nuclear weapons are an isotope of plutonium, plutonium-239, and an isotope of uranium, uranium-235. It is quite infamous for being used in Little Boy and other nuclear fission weapons. In nuclear weapons, the fission energy is released all at once to produce a violent explosion. In this case, uranium-235 is used as the nuclear fuel and its fission is triggered by the absorption of a slow moving thermal neutron. Despite this, it is also useful in uranium fuel. Models of the process suggested sustained fission reactions over a period of about a million years during a time period about two billion years ago. When nuclear fission is used to generate electricity, it is referred to as nuclear power. Some samples were found with a U-235 concentration as low as 0.29%. Also, fission-produced isotopes of neodynium and samarium were found. This indicated that some of the U-235 had undergone spontaneous nuclear fission at some point in the past. for nuclear weapons and nuclear power is its ability to fission. The most probable and radioactive product is iodine-131 (half life 8 days). In what is now Gabon in west Africa in 1972, French researchers found a deposit of uranium which had only 0.44% U-235 compared to the normal 0.72%. Uranium-238, the most prevalent isotope in uranium ore, has a half-life of about 4.5. Neutron Induced Fission - When an uranium-235 nucleus captures a neutron. Nuclear Fission Concepts The natural fission reactor: Gabon, Africa
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
